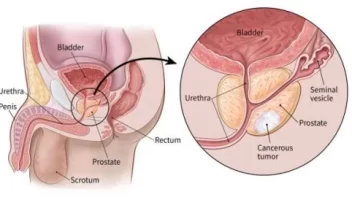
the introduction
In the era of personalized medicine, dealing with disease is no longer cancer It relies solely on clinical diagnosis. Advances in genomics and genetics have revolutionized the field, making genetic fingerprinting, or cancer genetic testing, one of the most important and advanced medical techniques in the diagnosis and treatment of tumors. This test relies on highly precise DNA analysis to detect genetic mutations responsible for cancer development or that increase the risk of developing it, thus opening the door to more effective and personalized treatment strategies.
The importance of this test today lies in its crucial role in guiding accurate diagnosis, determining the most appropriate treatment for each individual patient, and predicting the tumor's response to treatment. This has made its use an essential part of global protocols in modern oncology. This article is your comprehensive guide to understanding what genetic testing is, its advanced techniques, the differences between its types, and its pivotal role in improving cancer patient outcomes.

What is genetic fingerprinting (genetic analysis of tumors)?
Genetic fingerprinting, or **tumor genetic analysis**, is an advanced analysis that aims to study genetic mutations present within cancer cells or in healthy cells of the body. These mutations guide tumor growth and spread and identify its vulnerabilities. These genetic mutations can be classified into two main types:
Inherited Mutations:
These are mutations passed down from a parent and found in every cell of the body. These mutations do not necessarily mean cancer will develop, but they significantly increase the risk of developing cancer. The most common examples are mutations in the BRCA1 and BRCA2 genes, which are strongly linked to an increased risk of breast, ovarian, prostate, and pancreatic cancers.
Somatic Mutations:
These are mutations that occur in the DNA of a single cell or a group of cells during a person's lifetime as a result of exposure to a range of environmental or internal factors. These factors include exposure to smoking, chemicals, ultraviolet radiation, or errors that occur during cell division. These mutations are what cause tumor development and are the primary target of targeted cancer therapy.
Genetic testing aims to identify these mutations, whether inherited or acquired, to provide an accurate map of the tumor.
Main goals of genetic testing for cancer
The goal of genetic testing goes beyond traditional diagnosis, providing crucial insights that impact the entire treatment plan. Key objectives of **cancer genetic testing** include:
1. Targeting Therapy:
- **Choosing Targeted Therapy:** Testing identifies genetic mutations that can be targeted with specific drugs to stop the growth of cancer cells without harming healthy cells. For example, detecting an EGFR mutation in lung cancer guides the doctor to use EGFR inhibitors.
- **Determining the response to immunotherapy:** Certain mutations or markers of genomic instability (such as TMB and MSI) determine whether a patient will benefit from immunotherapy or not.
2. Predicting tumor behavior (Prognosis):
- **Predicting the future course of the disease:** The examination helps in predicting the behavior of the tumor, whether it will be aggressive and rapidly spreading or slow-growing.
- **Predicting patient response to medications:** This helps in determining the likelihood of a patient responding to chemotherapy or radiation therapy, thus reducing unnecessary treatments and associated side effects.
3. Screening for hereditary cancer risk:
- **Distinguishing between hereditary and non-hereditary cancer:** The examination helps determine whether the infection is caused by a genetic mutation that can be passed on to family members.
- **Risk Management:** Allows at-risk relatives to take preventive measures, such as frequent early screening or preventive surgery.
Detailed steps for conducting a genetic fingerprint test
The process of **genetic fingerprinting** is complex and precise, and it takes place in specialized laboratories. Although the sample collection method varies, the laboratory process follows standardized steps:
1. Sample collection and quality: The sample is the basis of accuracy.
The sample is usually collected in two main ways:
- **Tissue Biopsy:** This is the most common and preferred method for primary accuracy. A small sample is taken directly from the tumor, and this sample is used for direct genetic analysis of the cancer cells.
- **Liquid Biopsy:** Used in the analysis of circulating free DNA (cfDNA) or circulating tumor DNA (ctDNA).
2. DNA Extraction:
This stage is crucial to ensuring the quality of the analysis. DNA is separated from cells or from plasma (in the case of blood testing) using precise laboratory techniques and chemicals aimed at obtaining a sufficient and pure quantity of DNA.
3. Genetic analysis and the use of advanced NGS techniques
The accuracy of genetic testing depends on the technology used to read the genetic sequence. This is where modern technologies come into play:
Next Generation Sequencing (NGS) technology
NGS technology is considered the gold standard for comprehensive genetic analysis. This advanced technology allows for:**
- **Comprehensive Detection:** Detecting hundreds of genes at once in a single analysis (Gene Panels).
- **Speed and Accuracy:** Reading DNA sequences at high speed and at a lower cost than before.
- **Discovery of rare mutations:** Its ability to detect rare or new genetic mutations that may not be detected by older technologies.
Polymerase chain reaction (PCR) technology
It is used to detect specific, previously known mutations, such as one or two. It is fast and suitable for situations requiring a quick result or when the goal is to search for a predicted mutation.
4. Reading the results and interpreting the final report
After the sequencing process is complete, the massive amount of genetic data is analyzed using specialized computer programs (Bioinformatics).
- **Database Comparison:** The patient's genotype is compared to massive global datasets (such as COSMIC and ClinVar datasets) to determine the effect of the detected mutation on protein function and its relationship to the disease.
- **Interpretation of the final report:** This step is performed by the oncologist or geneticist. The detected mutations are linked to the **precise diagnosis**, the **targeted treatment plan**, and the **prognosis**.

The fundamental difference between blood gene testing (Liquid Biopsy) and tissue testing
Distinguishing between blood tests and tissue tests is an important aspect to understand when performing genetic analysis of tumors. Each has its role and advantages.
First: Blood test (Liquid Biopsy) – Non-invasive analysis
**Blood gene testing** is based on the analysis of circulating cancer cell DNA (ctDNA), which is released by dying or decaying cancer cells.
- **Key Features:**
- **Easy, quick and painless:** It does not require any surgical procedure or anesthesia, and is ideal for frail patients or those suffering from other illnesses.
- **Disease Progression Monitoring:** This is very useful for monitoring disease progression and response to treatment, as it can be performed repeatedly to detect the emergence of new resistance mutations.
- **Avoid difficult biopsies:** Ideal for patients who are difficult to biopsy, especially in cases of bone or brain tumors.
- **Tumor Heterogeneity Detection:** This shows changes in the tumor over time and in its various locations of spread.
- **Limitations (Defects):**
- It may not detect all mutations, especially if the amount of cancerous DNA is very small, as in early cancers.
- Less accurate in determining tumor characteristics in very early stages compared to tissue examination.
Second: Tissue Biopsy – The Gold Standard
**Tissue examination** is based on examining cancer cells directly from the primary tumor or metastases.
- **Key Features:**
- **The most accurate genetic test ever:** It provides the largest amount of tumor DNA, ensuring the highest accuracy.
- **Reveals the full characteristics of the tumor:** Suitable for comprehensive gene panel analysis and detection of complex markers such as fusion mutations that are difficult to detect in the blood.
- **Initial diagnosis:** This is the preferred method for the initial diagnosis of the disease.
- **Limitations (Defects):**
- It requires a minor surgical procedure (biopsy), which may cause pain or minor complications.
- Sometimes the sample may not be sufficient for full analysis (Insufficient Tumor Content).
- It cannot be easily repeated to monitor the response to treatment.
When is it advisable to undergo genetic fingerprinting?
Genetic testing has become a medical necessity in many cases, and is not limited to patients with advanced cancer. Testing is recommended in the following situations:
1. Suspected cases of hereditary cancer:
- **A strong family history of cancer:** Especially if the disease has appeared in several generations or in many family members.
- **The disease appears at a young age:** especially before the age of fifty.
- **Rare or multi-tissue cancers**.
2. Treatment planning for common cancers:
Genetic testing is essential before starting any targeted therapy, especially in cancers that studies have shown to be directly linked to targetable mutations:
- **Lung Cancer:** To detect mutations in *EGFR*, *ALK*, *ROS1* and others.
- **Colorectal Cancer:** To look for *KRAS*, *NRAS*, and *BRAF* mutations.
- **Breast Cancer:** To search for *HER2* and *BRCA* genetic and acquired mutations.
- **Skin Cancer (Melanoma):** To look for the *BRAF* mutation.
3. Cases of treatment resistance:
- **When the patient does not respond to conventional treatment:** If conventional chemotherapy or radiotherapy fails, genetic testing may guide the use of a new targeted treatment line.
- **In case of recurrence:** To reassess the mutations, the cancer cells may have developed a new resistance mutation.
Accuracy and reliability of genetic fingerprinting testing (Are the results accurate?)
The accuracy of genetic testing for cancer is among the highest in modern medical diagnosis. Physicians worldwide rely on it because it provides both quantitative and qualitative data on genetic changes, rather than solely depending on the phenotypic characteristics of cells.
NGS technology and reliability guarantee:
When using modern NGS technology, especially on high-quality tissue samples, the accuracy of mutation detection reaches excellent levels. Blood tests have also seen significant improvement in accuracy and are now reliably used for disease monitoring and early relapse detection.
The impact of accuracy on treatment decisions:
Large-scale studies indicate that genetic testing guides between 70% and 80% treatment decisions in modern cancers. This means that the decision to change the type of treatment, add targeted therapy, or enter a clinical trial depends directly on the results of this test.
In many cases, genetic testing is a direct cause of **improved treatment outcomes and increased recovery rates**, because it ensures that the patient receives treatment specifically designed for the mutations present in their tumor.
Key strengths and future role of genetic analysis
**Tumor Genetic Analysis** represents the driving force of Precision Medicine, and its strengths are evident in the following:
1. Enabling Targeted Therapy:
- It helps in selecting a precise treatment targeted to the specific gene mutation, which increases the effectiveness of the treatment and reduces severe side effects.
- It reduces unnecessary treatment that the patient will not benefit from, saving time, effort and resources.
2. Comprehensive disease monitoring:
- It helps in monitoring the disease and determining the likelihood of its recurrence long after treatment, especially when using periodic blood tests (Liquid Biopsy).
- It detects early hereditary cancers, allowing for preventive intervention.
3. Entering clinical trials (Clinical Trials):
Many promising new drugs in clinical trials rely on targeting specific gene mutations. Genetic testing results are key to qualifying patients for these trials, giving them access to cutting-edge treatments before they become commercially available.
4. Accurate Diagnosis:
It gives a more accurate diagnosis of the type of tumor and its molecular biological classification, a classification that has become more important than the traditional histological classification in many cases.
Challenges of genetic analysis and clinical interpretation
Despite the tremendous progress, the application of genetic testing faces some challenges that scientists and doctors are working to overcome:
1. Tumor heterogeneity:
Not all cells in a tumor are genetically identical. A biopsy taken from a single area may not represent the complete genetic picture of the tumor or its metastases. This is one of the reasons why liquid biopsy was developed to address this heterogeneity.
2. Mutations of unknown significance (VUS):
Sometimes, screening detects mutations whose clinical or pathological role has not yet been fully determined (Variants of Uncertain Significance). Interpreting these mutations requires considerable effort and monitoring of up-to-date global databases.
3. Cost and insurance coverage:
The cost of comprehensive genetic testing (NGS) can be high, and not all health insurance systems fully cover it, although the cost of ineffective treatment often exceeds the cost of genetic testing.
Summary and future prospects of genetic fingerprinting
**Genetic fingerprinting** is an essential and indispensable step in cancer diagnosis and treatment planning in the current decade. We have moved from a “one-size-fits-all” model to one of **precision personalized medicine**, where treatment decisions are driven by each patient’s unique genetic profile.
This advanced examination not only helps to accurately understand the nature of the tumor and select the most appropriate and effective treatment, but also directly contributes to improving the chances of recovery and reducing the burden of unnecessary treatment. As next-generation genetic testing (NGS) continues to develop and new generations of targeted therapies emerge, the pivotal role of tumor genetic analysis will continue to grow, becoming the greatest hope for patients and physicians in their fight against cancer.
If you or a family member are facing a cancer diagnosis, consulting an oncologist about undergoing comprehensive genetic testing is the first and most important step to ensure you receive the best and most accurate targeted treatment options available globally.